News

As a researcher, Kevin Kit Parker delights in exploring the nuances of cardiac cell biology and tissue engineering, traumatic brain injury, and micro- and nanotechnologies. An associate professor of biomedical engineering, he leads the Disease Biophysics Group at the School of Engineering and Applied Sciences, and he’s a core member of Harvard’s Wyss Institute for Biologically Inspired Engineering.
But he has another profession, one that’s equally important to him. He is a major in the U.S. Army, and late last summer he returned from his second tour of duty in Afghanistan.
There wasn’t much time for R&R. Parker’s lab needed his attention, and as keenly as he’d felt a sense of duty on the battlefield, he felt the same on campus. Almost a dozen manuscripts were nearing completion, a PhD student needed a dissertation ushered, and postdocs needed letters of recommendation.
“They did a great job while I was gone,” says Parker of his junior researchers, “but their lives can’t be put on hold for mine.”
The balancing act is a burden that would fall heavily on some, but Parker, known informally as Kit, is a larger-than-life figure (literally, at 6'5", as much as metaphorically) with an easy sense of humor and a deep Southern drawl that defuses tension even as it establishes leadership. Once you’ve been where he’s been, his manner suggests, there’s not much else to get riled about.
In a recent conversation, he talked about his work and drew connections between the lab and the battlefield.
What’s exciting you in the lab these days?
One thing is our tissue engineering effort. Two of my postdocs, Adam Feinberg and Mohammad Badrossamay, are working on new techniques to make protein textiles — making fabrics and fibers out of proteins. You can exploit the secondary and tertiary structures of the protein to get unique mechanical and chemical properties.
How did the work develop?
The nanofibers story is a good one. I got invited to speak at a Society of Laparoendoscopic Surgeons meeting on the future of surgery. I got there early and was checking out the equipment demonstrations, and it dawned on me: Wow, we do all this tissue engineering that starts off with a chunk of polymer. But now, when you get operated on, it all goes in through a small hole. Nothing we’re building in the lab right now is going to go into a hole that small.
So what did you do?
A long time ago, I’d seen someone use a cotton candy machine to weave fibers. I got this idea: What if you could take a cotton candy machine, weave some protein fabrics out of it, and then miniaturize it and put it inside the body? You’d go in there, inflate someone’s abdomen with gas, weave a three-dimensional structure with your nanofibers right in the body, and use it as a scaffold for your engineered tissue or to repair a damaged organ.
I had my lab manager, Josh Goss, go out and buy a cotton candy machine, and I just said, “Play with it. Just play.” Adam had already been doing protein nanofabrics, and I wanted to see if we could make something more deployable. So I hired Mohammad, who had a background in textiles and an in-depth understanding of polymer science. He got here two months before my last deployment to Afghanistan. I said, “Make me nanofibers. I gotta go fight.” And six months later, he had it. Josh is an expert scientific instrumentalist, and he essentially built our own version of a cotton candy machine — but it’s tricked out. Now we’re getting ready to start working with Rob Howe [Gordon McKay Professor of Engineering at SEAS], a surgical roboticist, to miniaturize this thing and take it in vivo.
What else are you working on?
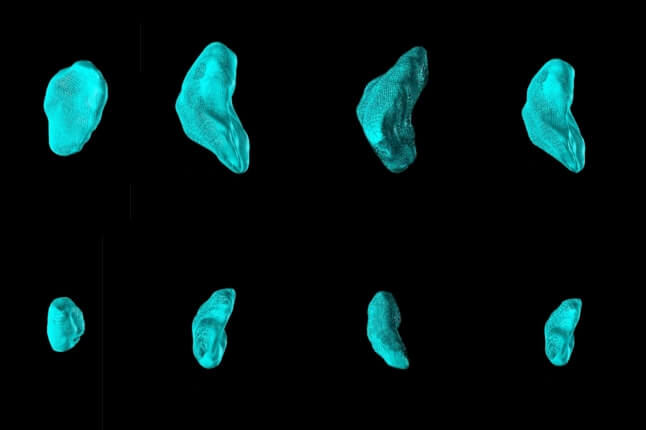
Our lab is primarily a cardiac tissue engineering lab, but we launched a big effort in traumatic brain injury [TBI] a few years ago. As an undergraduate in our lab, Borna Dabiri [AB ’07] started building tools for mimicking blast injury to engineered neurons and vascular tissues. He went to medical school at UC Irvine, but I convinced him to take a sabbatical to come back. He’s here on a Pierce Fellowship to do a PhD in biomedical engineering. He’s working with another PhD student, Matt Hemphill, and they’re going great guns after this TBI problem.
What’s their approach?
The idea is to build in vitro models of blast injury — to develop a set of tools that mainstream this science. Because in trauma science, the problem is that the experiments usually require animals. They’re pretty gnarly. The question is, can you use engineered tissues to mimic in vivo structures and do these kinds of experiments without blowing anything up. Our team has built a series of technologies to help us understand the scaling laws of blast injuries — understand them all the way from the protein ensemble or macromolecular scale up to whole engineered tissues.
It’s important work, judging from all the news about IEDs.
important work, judging from all the news about IEDs.
Even before it got out in the press, it was clear we had a problem. These guys were getting their bells rung. Early on, the IEDs were kind of crude, but on this last tour, I saw much more sophisticated technology. We’ve got soldiers out there that have been blown up 12 or 13 times. The question is, what are the long-term consequences for
these young guys? Are we going to see our VA hospitals flooded with a bunch of 40- year-old men who suffer from dementia, Alzheimer’s, Parkinson’s? This is a publichealthcrisis that we’re about 15 years out from, but it’s going to hit hard.
I believe there are therapeutic opportunities in first 10 minutes to an hour after a blast. What I want to know is, what are the signaling pathways that get turned on first? In combat casualty care, we talk about the golden hour, trying to get casualties off the battlefield and back to medical care within an hour. Now we’re starting to think about the platinum ten, the ten minutes after the injury, and about what’s happening. That’s when people are suffering a cascade of injuries that later can’t be reversed.
What part of your scientist self goes with you to the battlefield?
The scientific method is applicable anywhere. My first tour in Afghanistan, I had a very good team sergeant named Aaron Chapman. He and I would patrol and develop hypotheses about what we were seeing. We’d say, if this is what we think is happening, then we need to answers questions A, B, and C to test that out. The tools are different, the scale is different, the thinking is the same.
What part of the battlefield do you take back to the lab?
Managing people is probably the best thing I bring back. In both situations, the key to productivity is accountability and teamwork. Look, in both of my professions, the 18–30 year old demographic is where I live. I put my life in their hands on the battlefield, and I make my living with them in the lab. In both situations, these are young people who are pushing hard to push the envelope. I have seen what they’re capable of in the lab, and I’ve seen what they’re capable of on the battlefield. The achievers are the same, it’s just the medium of their achievement that differs.
Topics: Bioengineering
Cutting-edge science delivered direct to your inbox.
Join the Harvard SEAS mailing list.
Scientist Profiles
Kit Parker
Tarr Family Professor of Bioengineering and Applied Physics