News
PionEar team members Aaron Remenschneider, Nicole Black, Ida Pavlichenko, and Elliott Kozin are using materials science technology to design ear tubes that do not clog as easily and can improve drug delivery to the middle ear. (Photo courtesy of PionEar.)
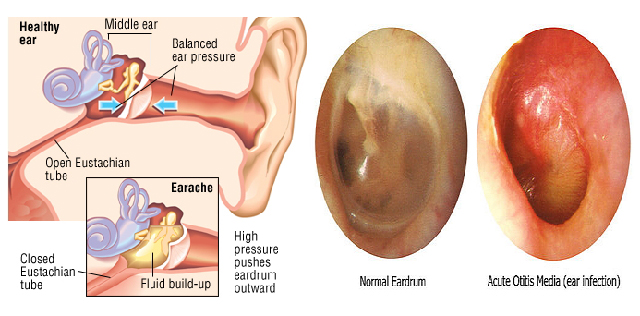
More than 700,000 children in the U.S. receive tympanostomy tubes each year to treat hearing loss and chronic ear infections, making it the most common pediatric surgical procedure. But a significant number of those tubes fail, leading to painful complications, invasive treatments, and the need for additional surgeries.
A collaboration among investigators at Harvard John A. Paulson School of Engineering and Applied Sciences (SEAS) and Massachusetts Eye and Ear aims to use advances in materials science technology to develop better tympanostomy tubes. On April 12, the team was announced as one of five finalists in the Health or Life Sciences track of the Harvard President’s Innovation Challenge as team PionEar. They will compete for a portion of $310,000 in seed funding, which will be announced at the award ceremony on May 2nd.
The idea of improving tympanostomy tube technology began when SEAS graduate student Nicole Black took a clinical otolaryngology class at the Massachusetts Eye and Ear. When classmates looked inside her ear, they noticed scar tissue from when she had ear tubes as a child.
“I started thinking, the scar tissue on my eardrum is probably not helping sound conduction. There has to be a better way to treat chronic ear infections,” said Black, a graduate research fellow in the lab of Jennifer A. Lewis, Hansjorg Wyss Professor of Biologically Inspired Engineering. “That was the starting point for me to think of this project in a different way, in that this is actually something that could potentially improve the lives of millions of patients.”
Black joined forces with Ida Pavlichenko, a postdoctoral fellow in the lab of another SEAS researcher, Joanna Aizenberg, Amy Smith Berylson Professor of Materials Science and Professor of Chemistry & Chemical Biology. Pavlichenko, herself the mother of a one-year-old who suffered from multiple ear infections, was eager to explore new advances in material science stemming from the Aizenberg lab that could be applied to improve ear tubes.
The foundation of a multidisiplinary collaboration was set in motion about a year ago when Pavlichenko and Black met with ear surgeons Aaron Remenschneider, Investigator at Eaton Peabody Laboratories and Elliott Kozin, Chief Resident in the Department of Otolaryngology at Harvard Medical School, to discuss how novel technology could improve patient outcomes.
“As soon as Nicole and Ida discussed their research on slippery surfaces, one obvious application arose,” Remenschneider said. “As surgeons, we spend a lot of time putting in ear tubes, but many limitations with the technology remain. Tympanostomy tubes in children often become clogged with cellular debris, which requires them to be replaced through a revision surgery under anesthesia. In addition, tubes may extrude too quickly or remain in the ear drum for an extended period of time, also requiring an additional procedure.”
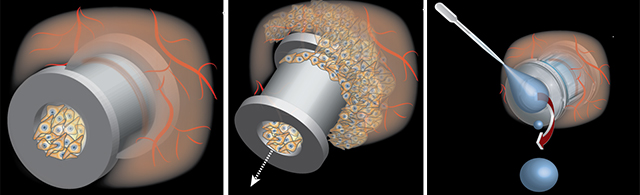
Often, tympanostomy tubes become clogged (left) or may extrude too quickly (middle). Current designs also offer limited ability to treat ear infections topically (right.) (Graphic courtesy of PionEar.)
“We discussed how one could harness the amazing advances in material sciences developed in the Lewis and Aizenberg labs and apply these technologies to tympanostomy tubes. Our goal is to improve tube performance and enable enhanced drug delivery to the ear,” Kozin said.
The goal of PionEar is to design ear tubes that do not clog as easily and can improve drug delivery to the middle ear. Current tubes have limited flow due to their small size, making it challenging to effectively treat infections with topical ear drops.
To solve these problems, the PionEar team is utilizing anti-fouling technology from the Aizenberg lab and novel materials from the Lewis lab to develop an easy-to-manufacture, liquid-infused ear tube that prevents cells from sticking to the surface. Their prototypes are smaller and easier to implant than current models, Black said.
As they fine-tune the design, the biggest challenges involve demonstrating the benefits of their technology, not only for young patients, but also for surgeons, hospitals, and insurers. The researchers will soon begin moving through the complicated FDA regulatory process.
“You can imagine how challenging it can be to be on the front line of developing a medical device,” Pavlichencko said. “We are currently in a continuous review process, trying to get as many opinions as we can about the top properties we should prioritize when we think about making a transition to using these tubes in patients.”
The team will also work with two students in the Harvard Summer Research in Global Health Program to identify global challenges. In developing nations, cost may be a bigger factor, or there may be different limitations on bathing or swimming. Seeing how their devices are viewed in a global context is an important consideration as the PionEar research team moves forward, Black said.
The team is taking advantage of the Harvard network, utilizing the resources of the Wyss Institute for Biologically Inspired Engineering, where both Lewis and Aizenberg are core faculty members, Harvard Innovation Labs Venture Incubation Program, and the Office of Technology Development to protect their intellectual property and develop a sound plan to reach human clinical trials.

The PionEar team is currently in the Venture Incubation Program at the Harvard Innovation Labs. (Photo courtesy of PionEar.)
While translating basic science research into an implantable device is new territory for the PionEar team, they are able to combine surgeons’ clinical expertise with bioengineers’ innovative mindsets to conceive novel solutions to a problem that has existed for decades, Kozin said.
“The goal of every surgeon-scientist is not only to treat the patient, but also to think about how he or she can improve the surgical management of patients by minimizing patient morbidity and creating novel therapeutics that may eliminate the need for surgery completely,” he said. “I think our project directly addresses these goals.”
For Pavlichenko, who knows firsthand the frustrations felt by parents of children who receive tympanostomy tubes, the opportunity to make an impact, even though a final product may still be several years away, is especially gratifying.
“I truly believe that we can bring a real product to patients, a product that could help children and millions of parents like me,” she said. “The opportunity to solve this problem is very inspirational.”

Topics: Bioengineering
Cutting-edge science delivered direct to your inbox.
Join the Harvard SEAS mailing list.
Scientist Profiles
Joanna Aizenberg
Amy Smith Berylson Professor of Materials Science and Professor of Chemistry & Chemical Biology
Press Contact
Adam Zewe | 617-496-5878 | azewe@seas.harvard.edu




